By Edward Milner and Ana Suarez-Suarez
Postdoctoral Researcher Dr Edward Milner, working on the NERC-funded MeteoRR project, visited the world leading research group of Professor Korneel Rabaey at the Center for Microbial Ecology and Technology at the University of Gent in Belgium. Edward has been working on the use of bioelectrochemical systems (BES) to recover metals from wastewaters. He was fortunate to learn first-hand from Professor Rabaey and his group about their exciting new research in this area and practical details for setting up novel BES related to his work on the MeteoRR project.
Bioelectrochemical systems (BES) explained
 BES technology can be used for resource recovery from wastewater. They can simultaneously remove organic contaminants and produce electricity, valuable chemicals or recover metals. Additionally, BES are being adapted with the potential to convert the greenhouse gas CO2 to valuable organic feedstock chemicals.
BES technology can be used for resource recovery from wastewater. They can simultaneously remove organic contaminants and produce electricity, valuable chemicals or recover metals. Additionally, BES are being adapted with the potential to convert the greenhouse gas CO2 to valuable organic feedstock chemicals.
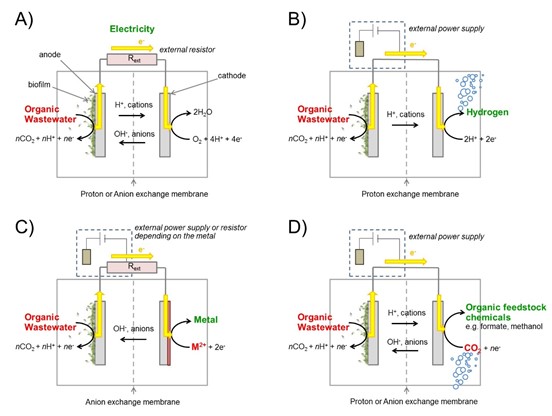
In BES, wastewater containing organic chemicals is fed into an anode chamber where the organics are broken down by specialised bacteria into CO2, protons and electrons. In the process, the electrons are released into the anode electrode where they move through an external circuit to the cathode electrode. In a microbial fuel cell, these electrons combine with oxygen at the cathode, and electricity is generated through the movement of electrons in the external circuit. Alternatively, the electrons can be combined with protons at the cathode to produce hydrogen in a microbial electrolysis cell, metal ions to recover pure metals, or CO2 to produce valuable organic feedstock chemicals such as formate or methanol.
In a microbial fuel cell, the system produces electricity whilst in a microbial electrolysis cell. The system produces hydrogen but requires a small amount of additional electrical energy provided by an external electrical power supply. Similarly, additional electrical energy is required for CO2 conversion to valuable organic feedstock chemicals. For metal recovery the required additional electrical energy depends on the metal being processed. The different cathode reactions in BES may require chemical or biological catalysts at the cathode to make them work.
Environmental and economic benefits of BES
Newcastle University leads two major research projects focused on BES: The MeteoRR project led by Professors Ian Head and Tom Curtis in the school of Civil Engineering and Geosciences, and the LifesCO2R project led by Dr Eileen Yu and Professor Keith Scott in the school of Chemical Engineering and Advanced Materials.
The projects develop BES for the recovery of pure metals or valuable chemicals with market value from wastewater containing organic, metal and CO2 pollutants. This offers economic incentives for industry to adopt clean technologies as part of the circular economy.
BES technology can treat wastewaters by removing organic and metal pollutants which are harmful to living organisms, including people, in the environment. Untreated wastewater containing organics promotes microbial pathogens in the environment which can cause disease in people, whilst wastewater containing heavy metals can be toxic to human health. These wastewaters can cause adverse effects on fragile ecosystems. Additionally, using BES to remove CO2 and recover valuable organic feedstock chemicals has the additional environmental benefit of helping to combat global climate change caused by greenhouse gas emissions.
Read more about BES technology and the environmental and economic benefits in MeteoRR’s articles and reports.
MeteoRR is a NERC-funded project within the Resource Recovery from Waste programme. The project has teams at the universities of Newcastle, Manchester, Surrey and South Wales, and formal collaborations with researchers at Glasgow, Penn State, Ghent and Harbin. The teams work closely with industrial partners who will benefit from the research findings.
This article was originally published on LinkedIn on 9th January 2017.

